Apple Blotch Disease
ID
SPES-509NP
Introduction
Apple blotch (also known as Marssonina leaf blotch) is an emerging disease in the Mid-Atlantic United States. It is caused by the fungus Diplocarpon coronariae (syn. Marssonina coronaria). This disease leads to severe defoliation that indirectly affects the apple fruit size, color, yield and twig development. Apple blotch disease is known to cause serious losses in Asia and Europe, especially on apple scab resistant cultivars. In the United States, it was first reported in Wisconsin (Davis 1903) but was never considered a significant disease before 2017. However, excessive rain during the summer of 2017 in the Mid-Atlantic United States favored outbreaks of apple blotch disease and has caused severe defoliation before harvest. Orchards with reduced fungicide spray programs were severely affected.
Symptoms
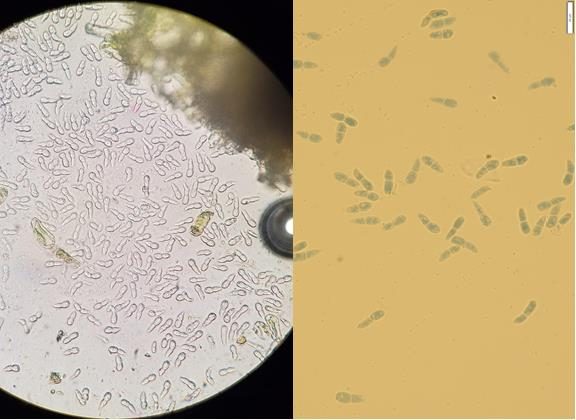
Apple blotch disease first starts with symptoms of small circular, purple to brown spots on the surface of the leaves (Fig 1a, b), within which small black asexual fruiting bodies called acervuli form. These bodies are visible as tiny black dots and produce asexual spores called conidia (Fig. 2). With time, smaller leaf spots enlarge, get light brown center with purple edge, and eventually develop to larger brown blotches with margins showing fungal mycelium strands (Fig. 1c). These blotches coalesce, cause yellowing or browning of leaves and lead to premature defoliation. Leaf infections provide inoculum for infections of fruit, which appear as black, concave spots with light-brown center (Fig. 1d). Depending on the cultivar, yellowing surrounding the leaf blotches might or might not appear (Fig. 1c). Severe crown defoliation can reduce marketable yield, affect fruit quality and reduce the overall vigor of apple trees unless fungicides are applied.

Disease Cycle
The life cycle of this disease starts in the spring with spores released by rain splash and aerosol from overwintered leaves on the orchard floor. Spore dissemination and infections occur during or after long periods of rainfall at temperatures from 20 – 25°C (68 – 77°F). Once spores reach the leaves, they germinate into an infection hypha and penetrate the cuticle of the leaf. After incubation, leaf spots become visible and acervuli form on the leaf surface. If the weather conditions are conducive, defoliation can start as early as 2 weeks after the first spot symptoms appear. First leaf spots usually appear between June to July in the northern hemisphere.
Control Options
Apple blotch disease is managed by orchard sanitation, cultural practices such as pruning, and timely fungicide applications.
Cultural Control
The incidence of apple blotch disease can be reduced by orchard sanitation and cultural practices. Shredding the fallen leaves on the orchard floor which serve as sources of inoculum, by flail mower or raking and burning the leaves can reduce the spore dose next spring. Dormant pruning should modify the tree crown to allow good air circulation inside the canopy and rapid foliar drying after rainfall or dew. This diminishes conducive microclimate conditions inside and between the trees for infections and allows better fungicide coverage increasing their efficacy. Application of 40 lbs of urea/A in 100 gals of water onto the overwinter leaves and raking the leaves from under the trees for later shredding by flail mower can reduce the inoculum level of this disease. Avoid planting highly susceptible cultivars such as ‘Rome’, ‘Mutsu’, ‘Royal Gala’, and ‘Golden Delicious’.
Organic Control
Application of dolomitic lime (2.5 tons per acre) just before leaf drop in fall or early in the winter can help apple leaf litter breakdown and thus aids to reduction of the spore inoculum dose in the overwintering leaves (this rate can also be used in apple orchards with synthetic fungicide programs). During the season, 10 – 12 spray applications per year of Funguran (copper hydroxide), sulfur, or lime sulfur can provide good control of leaf infections.
Chemical Control
Fungicides applied during the late spring and summer for control of other diseases like Sooty Blotch and Flyspeck and apple fruit rots are usually effective in controlling this disease. The first fungicide labeled for apple blotch control in the United States is Cevya (EPA Reg. No. 7969-407) with the active ingredient mefentrifluconazole (FRAC Group 3). However, if the fungicide applications are not delivered in intervals dictated by the frequency and amount of rain events, their efficacy can fail (fungicides are ineffective after 2 inches of rain occurring in a single or several wetting events). Whenever possible, fungicides should be integrated with the aforementioned cultural control practices. Fungicide applications are critical to protect leaves from this disease and prevent production of spores on them. Rotation of the most effective fungicides with different modes of action in FRAC Groups 3, 7, or 9 is recommended to prevent fungicide resistance that has been reported in other countries.
Germplasm Resistance
Several resistant cultivars have been bred in China, such as Yepingguo 9 and Dianchihaitang. Currently, in the U.S.A., there are no breeding programs that are developing cultivars resistant to apple blotch disease. However, cultivars like ‘Granny Smith’, ‘Pink Lady’, and ‘Gibson’s Golden’ are moderately resistant. If resistant cultivars become available in the future, they would be one of the key strategies for the integrated management of apple blotch disease.
Disease Infection Prediction
The infections by D. coronariae can be predicted by using a RIMpro Marssonina coronaria model (RIMpro B.V., Amsterdam, Netherlands), available by subscription at: https://www.rimpro.eu/. The model can use the U.S. National Weather Service weather forecast and can be connected to the local weather stations in the Network for Environment and Weather Applications (NEWA) available at: https://newa.cornell.edu/, to allow accurate model infection predictions and thus guide timely fungicide applications.
References
Aćimović, S. G. (2023). (A) Urea/lime for reducing apple scab and apple blotch disease overwintering inoculum; (b). Delayed dormant copper for apple, pear & peach, accessed 19 April 2023, Available online at: https://treefruitpathology.spes.vt.edu/2023/02/11/a-urea-lime-for-reducing-apple-scab-and-apple-blotch-disease-overwintering-inoculum-b-delayed-dormant-copper-for-apple-pear-peach/.
Aćimović, S. G., and Donahue, D. J. 2018. Marssonina leaf blotch of apple - A growing problem in South-Eastern New York. Tree Fruit News. 6:1-4.
Dang, J. L., Gleason, M. L., Niu, C. K., Liu, X., Guo, Y. Z., Zhang, R., et al. 2016. Effects of Fungicides and Spray Application Interval on Controlling Marssonina Blotch of Apple in the Loess Plateau Region of China. Plant Disease, 101:568-575.
FRAC 2022 - FRAC Code List 2022: Fungal control agents sorted by cross-resistance pattern and mode of action (including coding for FRAC Groups on product labels). Available online at: https://www.frac.info/docs/default-source/publications/frac-code-list/frac-code-list-2022--final.pdf?sfvrsn=b6024e9a_2
Khodadadi F., Martin P. L., Donahue D. J., Peter K. A., Aćimović S. G. (2022): Characterizations of an Emerging Disease: Apple Blotch Caused by Diplocarpon coronariae (syn. Marssonina coronaria) in the Mid-Atlantic United States. Plant Disease, 106(7): 1803-1817.
Khodadadi, F., González, J. B., Martin, P. L., Giroux, E., Bilodeau, G. J., Peter, K. A., ... & Aćimović, S. G. (2020). Identification and characterization of Colletotrichum species causing apple bitter rot in New York and description of C. noveboracense sp. nov. Scientific Reports, 10(1), 11043.
Khodadadi, F., Peter, K. A., Donahue, D., & Aćimović, S. G. (2019). Marssonina blotch of apple caused by Marssonina coronaria in New York. Plant Health 2019, 3-7 Aug, APS Annual Meeting, Cleveland OH.
Li, Y., Hirst, P. M., Wan, Y., Liu, Y., Zhou, Q., Gao, H., ... & Han, M. (2012). Resistance to Marssonina coronaria and Alternaria alternata apple pathotype in the major apple cultivars and rootstocks used in China. HortScience, 47(9), 1241-1244.
Marssonina Blotch of Apple (Online article), accessed 14 February 2023, https://plantix.net/en/library/plant-diseases/100341/marssonina-blotch-of-apple.
Marssonina Blotch on Apple Trees, University of Missouri, accessed 14 February 2023, https://ipm.missouri.edu/MEG/2021/4/marsonin a-MW/.
Marssonina Leaf Spot, Montana State University, accessed 14 February 2023, https://www.montana.edu/extension/pubs/a-guide-to-pests-problems-and-identification-of- ornamental-shrubs-and-trees-in-montana/diseases/marssonina-leaf-spot.html/.
Marssonina: Is this a new pathogen? By Matt Milkovich, Good Fruit Grower, March 10, 2021. Available online and accessed 14 February 2023, https://www.goodfruit.com/marssonina-is-this- a-new-pathogen/.
Oberhänsli T., Dalbosco A., Leschenne V, Bohr A., Buchleither S., Tamm L., Wille L, Aćimović S. G., Khodadadi F., Ryu Y-H., Studer B., Schärer H-J., Patocchi A. (2021). Multiplexed SSR marker analysis of Diplocarpon coronariae reveals clonality within samples from Middle Europe and genetic distance from Asian and North American isolates. CABI Agriculture and Bioscience, 2, 21.
Peter, K. A. (2018). Apple Disease - Marssonina Blotch. Penn State Extension. Available online at: https://extension.psu.edu/apple-disease-marssonina-blotch.
Peter, K. A. (2023). 2023 Disease Update: Disease Conditions Favored for Marssonina Blotch and Apple Scab April 28–May 1. Available online at: https://extension.psu.edu/2023-disease-update-disease-conditions-favored-for-marssonina-blotch-and-apple-scab-april-28-may-1
Sagong, D. H., Kweon, H. J., Song, Y. Y., Park, M. Y., Nam, J. C., Kang, S. B., & Lee, S. G. (2011). Influence of defoliation by Marssonina blotch on vegetative growth and fruit quality in 'Fuji'/M. 9 apple tree. Horticultural Science & Technology, 29(6), 531-538.
Sharma, J. N. (2000). Marssonina blotch-a new disease of apple and its control. Indian Journal of Plant Protection, 28(1), 100-101.
Sharma, J. N., Sharma, A., & Sharma, P. (2003, October). Out-break of Marssonina blotch in warmer climates causing premature leaf fall problem of apple and its management. In VII International Symposium on Temperate Zone Fruits in the Tropics and Subtropics 662 (pp. 405-409).
Takahashi, S., Sawamura, K., & Sato, Y. (1990). Marssonina blotch. Compendium of apple and pear diseases, 2nd Edition, ed by Sutton T. B. et al., APS Press, St. Paul, MN. 46-47.
Virginia Apples, accessed 19 April 2023, https://www.virginiaapples.net/apple-varieties.
Yin, L., Li, M., Ke, X., Li, C., Zou, Y., Liang, D., & Ma, F. (2013). Evaluation of Malus germplasm resistance to marssonina apple blotch. European journal of plant pathology, 136, 597-602.
Wöhner, T., & Emeriewen, O. F. (2019). Apple blotch disease (Marssonina coronaria (Ellis & Davis) Davis)–review and research prospects. European journal of plant pathology, 153(3), 657-669.
Virginia Cooperative Extension materials are available for public use, reprint, or citation without further permission, provided the use includes credit to the author and to Virginia Cooperative Extension, Virginia Tech, and Virginia State University.
Virginia Cooperative Extension is a partnership of Virginia Tech, Virginia State University, the U.S. Department of Agriculture (USDA), and local governments, and is an equal opportunity employer. For the full non-discrimination statement, please visit ext.vt.edu/accessibility.
Publication Date
June 14, 2023



