Small Scale Perennial Production Using Iris as an Example Crop
ID
SPES-354P
EXPERT REVIEWED
Note: Terms defined in the glossary are italicized on first use in the text.
Introduction
Many ornamental crops are produced by breeders, hobbyists, and garden enthusiasts through small scale operations. This publication covers key components of small scale in-ground production of perennials, including site evaluation, infrastructure, setup, production, pest management, and marketing. Information is based on production methods for a 1/8 acre bearded (Iris germanica) and beardless (Iris hexagona and Iris siberica) iris operation. Both types of irises are distinguishable from each other by the presence or absence, respectively, of a fuzzy appendage referred to as a “beard” which occurs on the lower horizontally held falls (sepals) of the bloom. Their anatomical position distinguishes them from their vertically held counterparts, the true petals, referred to as standards. Although irises are used as an example throughout this publication, the information presented is relevant for all types of perennials grown on a small scale.
Site Assessment
A thorough assessment should be conducted to determine if a site is suitable for production. Making a checklist from the questions below can be useful for gathering information to help make informed decisions.
Area
Do zoning ordinances allow for a commercial business?
Is the land open/clear enough for any necessary buildings or structures?
Sun/shade
Is there sufficient space for production areas in full sun, part sun or full shade for the kinds of plants that will be grown?
Topography
What is the topography of the site? Is there a topographic map available?
Are there slopes that would impact production areas, stormwater runoff (erosion), buildings, and foot or vehicular traffic?
Can the topography be used to collect runoff as a source for future irrigation needs in order to control costs?
Soil condition
Has there been a recent basic soil test? This would determine soil pH and nutrient levels which impact in-ground production.
Is the soil compacted? This could cause drainage problems and erosion, and the ground could require aeration and/or amending to correct the problem.
Is the soil consistent across the property? Soil conditions can vary greatly across an area depending on the history of the property.
Wildlife
What are the potential wildlife conflicts? Deer, geese, racoons, and voles can cause significant damage to production areas and crops.
What would managing unwanted wildlife entail?
Water
What is the crop’s irrigation need?
• Irrigation is generally recommended in order to produce uniform plants quickly.
• An irrigation system that has different zones and that can be converted easily between drip and overhead should be considered for growing crops with different water needs at the same time or for differing water needs at different growing stages of a specific crop.
What will be the water source for irrigation?
• If using city water, the cost should be factored into the production costs.
• If using well water, it should be tested for quality and irrigation suitability. Agricultural labs that test soil often test water, too. Well water can have a combination of high salts, total alkalinity, or iron that can discolor, stunt or even kill plants.
• Is there an existing pond on the site, or an opportunity to create a pond that could be used for stormwater collection and for irrigation? Are there any safety regulations about signage or fencing around the pond?
Materials and Equipment Considerations
Each operation will have unique materials and equipment requirements. The challenge is making appropriate choices which allow for profitable production and future growth of the operation. Acquisitions need to be carefully considered at all times to control expenses. Small scale operations have tight budgets. While some debt should be expected in the beginning when establishing an operation, eliminating any debt and turning a profit as quickly as possible is the goal.
Considerations include:
What does the operation need to start and function effectively at a minimal level?
• Raised bed materials.
• Growing media or soil amendments.
• Irrigation infrastructure (overhead, mist, drip), timers, rain sensors.
• Starter plant stock.
• Wildlife protection.
What activities will require specific equipment?
• What type of specific equipment is needed?
• Are there alternatives that would work — build your own, borrow, lease, buy used, cost-share with another producer?
• Will the amount of use justify the cost of the specialized equipment?
• Will the specialized equipment increase operational efficiency?
Can the operation currently afford this material or equipment expense?
Will this item address current needs, increase efficiency, and pay for itself quickly?
Possible materials and equipment for startup include:
Hand tools — shovels, rakes, etc.
Cultivator and/or tiller.
Weeding implements.
Growing media vs. existing soil.
Soil amendments.
Fertilizer and/or lime.
Framing materials for raised beds.
Irrigation system materials.
Wheelbarrow/cart.
Specific tools/equipment.
A website or marketing outlet.
Office — space, supplies, business software (accounting, inventory, sales), order fulfillment setup.
Once the site assessment has been accomplished and materials and equipment information gathered, then the design, layout, and implementation can begin.
Planting Area
Organizing and using production space efficiently is important to maximize crop production and profits. Production space can be organized as either raised beds, rows, or a combination of both. Any layout should be based on the site topography, crop requirements for optimum growth, and access considerations. Using basic equipment like stakes, survey string, flagging tape or paint, a tape measure or measuring wheel, hammer, and calculator, the production area can be laid out very easily and efficiently (Niemiera 2018, Schrock 1994). Figure 1 shows how to establish a temporary layout that can be used to test bed access and traffic flow patterns for people and equipment before the permanent installation.

Raised Beds
Raised beds create infrastructure above ground when the natural soil cannot be used due to compaction, poor nutrients, poor drainage, debris or roots, etc. (Schrock 1994).
A raised bed’s height is usually established as 6 or 8 inches (the standard widths of treated boards), but it can be whatever is practical from readily available materials. Landscape timbers, bricks, pavers, or retaining wall stones are all possible materials to use. The frame height will impact the volume of growing media needed to fill the bed. Beds on slopes might only need the downhill side wall to contain growing media. Seating the frame level and securely on the ground is important to prevent loss of growing media from under the frame edges (Schrock 1994).
One- to three-sided raised beds can work fine on slopes to create a terraced appearance while four-sided beds are used on flatter terrain. The side walls of beds on slopes can be tapered to fit the slope angle rather than excavating the hillside (figure 2).

Bed construction can be expensive. It should be based on the minimum number and size of the beds required to keep expenses down, expanding only based on operational demands. Using materials already on site or repurposed/recycled materials can keep these costs manageable.
Rows
Planting rows can be used alone or in combination with raised beds. Irises and many other perennials can be grown quite well this way (figure 3). The same layout process as for raised beds can be applied. Rows should run perpendicular to or across slopes, not parallel to the slope, to prevent them from creating channels for runoff and erosion (Weil and Brady 2017).

Access
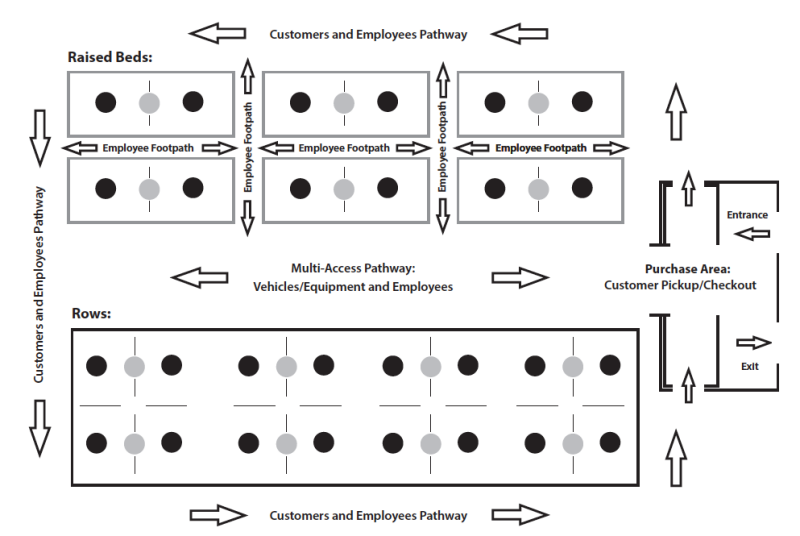
When designing a production space layout, include access space in accordance with traffic flow requirements (figure 4). It is important to consider who and what will be moving around the site along with where and to what they need access. Will only employees need access or will customers and tours need to be accommodated? Does there need to be separation of foot and vehicular traffic for safety/operational concerns? What type of equipment will be used — wheelbarrows, pull carts, golf carts, ATVs, or cars and trucks? Pathway width, traffic-flow direction, surface material, edging, and maintenance should all be considered. Pathways should be maintained as stable and passable regardless of type of traffic. Equipment selection and automated implements can reduce the amount of land needed for access space, allowing more area to be used for production, thus increasing the plants per unit area (Schreiner’s, n.d.). Figure 4 is an example of an access pathway layout for an operation with both raised beds and rows and different types of traffic.
Growing Media and Nutrients
Testing of soil (or growing media in raised beds) should be done every one to four years or as needed based on considerations such as media composition, plant growth issues related to nutrient deficiencies, adding amendments, renovating old beds, or installing new beds (Freeborn and Relf 2017). Testing can be done independently in response to any of these considerations or as part of an annual nutrient management plan to determine if sufficient nutrients are available or if any corrections need to be made (PWSWCD, n.d., Freeborn and Relf 2017). Fostering healthy soil ecology by adding organic matter like plant- or manure-based compost to the soil or growing media will reduce expenditures on chemical fertilizers while establishing a proper nutrient balance over time (Weil and Brady 2017). Soil with regular additions of organic matter will develop structure and a healthy microbial biome, which supports healthy plant growth (Weil and Brady 2017, Freeborn and Relf 2017). Figure 5 shows growing media in a raised bed that was initially composed of a plant-based compost mixed with existing heavy clay soil. The bed received annual top dressings of organic matter for six years. Note the healthy plant growth and few weeds present.

Healthy soil or growing media should supply the necessary nutrients for plant growth. The soil pH scale goes from 1 to 14, with seven being neutral. Below seven is acidic and above is basic or alkaline. The optimal range for growing healthy plants is 5.5 to 7.5 (Freeborn and Relf 2017); where all essential nutrients are available if present. Some crops have specific growing conditions. Know the crop’s preferred conditions and manage production spaces accordingly. Irises prefer neutral to slightly acid growing conditions, around pH 6.1-7.2, according to the American Iris Society (AIS 2011). Bearded irises (e.g., I. germanica) prefer low nitrogen (N) levels with higher amounts of phosphorus (P) and potassium (K), while beardless irises (e.g., I. hexagona) do well with more even amounts of N, P and K. If testing shows that nutrients need to be added for a crop, then match the fertilizer to the crop. For example, bearded iris might need low N formulations such as 6-10-10 or 5-10-10 (N-P-K); while beardless irises might need more balanced formulations such as 8-8-8, 10-10-10 or 14-14-14 (Schreiner’s, n.d., AIS 2011). Fertilizers are applied twice annually according to label rates on the packaging, roughly four to six weeks prior to bloom and any time after bloom all the way up to early fall, regardless of the kind of irises being grown. Timing at the prescribed rate for the total growing area is the critical factor in the application, and all applications should be stopped two to three weeks prior to the first hard frost (AIS 2011). Choose the correct fertilizer and application method for the crop. Water soluble fertilizers can be mixed according to label rates and applied manually as a liquid drench or siphoned through the irrigation system. Granular applications can be broadcast over a bed, applied to an individual planting hole or row, or incorporated into a bed area. Granules can be quick release when dissolved by irrigation, rain, or soil moisture, or they can be controlled (slow) release over a three- to four-month growing season. Fertilizer selection and application rate and time will depend on the specific crop, soil or growing media analysis, growth stage of the crop, equipment needed, product availability and producer capability or preference.
Irrigation
Soil or growing media should be properly irrigated to provide consistent moisture for optimum crop production (Straw 2015). It is generally recommended that a rate of 1 inch of water per week be applied in the absence of natural rainfall to any sized production area, sufficient to wet soil to a depth of 5-6 inches (Straw 2015, SNA 2020). Each crop’s watering requirements are different and are influenced by the soil or growing media and crop stage of growth. For example, bearded irises, once established, do not require much additional water beyond what is provided by natural precipitation (barring a drought), while beardless irises prefer more frequent and consistent moisture levels (Schreiner’s, n.d.). Applying mulch can conserve water, reducing irrigation costs.
Consider the irrigation source carefully. Irrigating from a public water source costs money, which increases operational expenses (PWCSA, n.d.). Use of well water will require specific tests to determine if the water quality is appropriate for irrigation purposes. Nutrient and mineral levels in well water can be high enough to be toxic to plants and can affect the type and frequency of fertilizer used. The supply pressure and the number of wells needed to accommodate crop production should be determined early in the planning process (SNA 2013). Installing a well can also be expensive and significantly impact the budget. Irrigation water from a pond or collection tank/cistern can be used and should also be tested to determine quality. Filters will definitely be necessary on systems using well, tank, or pond water to prevent clogging of the irrigation heads or emitters.
Commercial irrigation companies have consultants and websites that can help with designing a system appropriate for a specific crop. It is better to work through a design on paper first before installing a system (Rainbird, n.d.; Orbit, n.d.; Dripdepot, n.d.; Dripworks, n.d.; Mister Landscaper n.d.). Systems can distribute water differently based on what is needed. Common types of irrigation systems are micro-drip, drip, and overhead micro spray, mist or sprinkler. Drip systems apply water at the soil surface which reduces evaporation and the overall amount of water applied (SNA 2013). Overhead systems apply water through the air at the base or over the top of the crop, which causes frequently wet foliage. Irrigation should be scheduled for early in the day, which allows time for the soil/growing media and plant foliage to dry throughout the day. This helps control diseases, which can thrive under moist conditions. Figure 6 shows components of a micro-spray emitter system that can be connected to a rainwater harvesting tank with a controller.

A rain sensor should be included in the irrigation design so the system will not run when there is sufficient natural rainfall. Overwatering can cause poor crop growth and encourage diseases.
Plant Stock
Take the time to learn about the different types of plant stock and find quality sources for them. Irises are generally sold as single bare root divisions from primary sources but can be found in pots from secondary sources as well. They can even be grown from seeds when dealing with species or wild-type varieties.
Stock Selection
Hybridizers generally have their own businesses or sell their irises directly through larger grower/hybridizer operations, which may offer stock at wholesale prices for buyers who need large quantities. Most horticultural and plant societies also have source lists. All three of these can be primary sources for high-quality, pest-free, and true-to-name stock. Secondary sources should only be used if the plants can be seen in bloom to confirm stock identity. The burden is on purchaser to know what they are buying since secondary sources may not know much about the plants. Stock plants may also be obtained at auctions, but the prices can be prohibitive.
There is a much smaller market for species irises, varieties, and those irises that have been on the market beyond 30 years (referred to as historic irises); which are still of some commercial value (Joe Pye Weed’s Gardens 2020, AIS 2018, HIPSb, n.d., BHIG, n.d.). These irises are generally sought only by hobbyists, collectors and conservationists for use as landscape plants. Species irises were useful in early hybridizing efforts during the 1800s to establish commercially viable modern hybrids that expressed better traits such as more vigorous growth, disease resistance, and larger, more colorful and abundant blooms. For buyers interested in these irises, the primary sources are specialty growers listed on iris society source lists (AIS 2018, HIPSa, n.d.). Small quantities may be found through secondary sources sold as potted perennials at market prices. Many irises are grown from seed which can cost little to acquire (SIGNA, n.d.).
Operators who are involved in both resale and hybridizing of plants should select stock that serve both purposes. Quality plant stocks exhibit both vigorous growth habits and interesting genetic traits, which support good sales volumes and allow for their use in hybridizing new, higher-priced introductions. Inferior stocks are those that do not perform well in these same characteristics. They should be removed from inventory to be composted or put into the trash and not utilized for sales or hybridizing. Diseased stock plants should be discarded in the trash and not utilized under any circumstances. Stock that does not suit hybridizing goals or has become commercially unviable but still grows well may be sold at a discount.
In some cases, plant patents affect which cultivars can be used as plant stock by commercial operations. Irises are not patented. New cultivars are registered by the hybridizer with The American Iris Society, which is the world repository for new iris registrations in a permanent cultivar database. The value of new cultivars falls so quickly within the first five years once introduced to commerce that the cost of patenting is prohibitive. Once acquired through normal commercial sales, irises are often traded among friends or passed down as family heirlooms. Iris cultivars may be used without any royalty payments or permissions.
Even a small scale iris producer should include hybridizer names, registration years, and introduction years with cultivar lists, especially if those details are not on a field label. Some cultivars of other perennials, such as daylilies, bought at commercial box-store-style nurseries may show a name tag with “PPAF” displayed, which indicates a Plant Patent has been Applied For. It is wise to avoid purchasing these and also to check with the registrar of the horticultural society specific to the plant stock to determine if new cultivars are patented and if there are any usage guidelines for production operations regarding sales of patented plants. Buyers can also check for patents with the United States Patent and Trademark Office (USPTO, n.d.). One unspoken courtesy appreciated by hybridizers is to check in with them on the appropriate sales prices for their introductions until market prices fall to commodity levels.
Primary Sources
• Growers and hybridizers with established reputations.
• Horticultural society plant sales.
• Horticultural society source lists.
Secondary Sources
• Plant auctions.
• Garden centers.
• Friend’s home gardens.
• Family hand-me-downs.
• Roadside plants.
• Unknown Cultivars with NO IDentity (NOID status) (HIPSb, n.d.).
• Stock acquired through extended supply chains of two or more sources
Pricing
Many methods can be used to arrive at a final price for a product; some involve aspects of market research and psychology (Kohls and Uhl 2015, Uva 2009). At the very least, the plant price should cover production costs with a markup for profit. Take time to research current market pricing and trends. Sales could include the resale of currently available cultivars and varieties, sales of newly hybridized introductions, or a combination of both. Table 1 shows how 2,000 square feet of bed space within a 1/8-acre area may be allocated as an iris production operation to support both resale and hybridizing functions along with expansion space for future growth of the operation. This table does not account for access space. Selling larger quantities of currently available cultivars at standard market prices is a different business model than selling smaller quantities of new cultivars at higher premium prices. The novelty of new introductions is what drives the high prices initially. Garden performance influences prices later as cultivars age and people share their gardening experiences. New introductions depreciate over an average 30-year commercial lifespan down to the standard commodity price ranges as shown in table 2. Market demand, cost of production, and commercial age of each cultivar ultimately determine when a plant is no longer commercially viable (worth producing). Each operation must make its own determination regarding this cutoff point.
| Plant Stock | Cultivar #’s (a) | Bed Space ft2 (b) | Ft2 / Cultivar (b/a) | ft2 % |
|---|---|---|---|---|
| Resale Plant Stock (1 to 4 starter plants of each cultivar) | 200 | 800 | 4 | 40 |
| Hybridizing (seedlings) | 150 | 600 | 4 | 30 |
| Expansion Space | TBD* | 600 | TBD* | 30 |
| Total Area | 2,000 | NA^ | 100 |
* TBD = to be determined. ^ NA = Not applicable.
| Iris Plant Stock | Average Market $ / Plant1 | Commercial Lifetime $ Range (30 years) |
|---|---|---|
| Depreciated Regular Stock: • Bearded • Siberian • Louisiana • Species/Varieties2 |
$5-$7 $6-$8 $8-$10 $10-$25 |
$75-$5 $35-$6 $50-$8 $25-$10 |
| First Year Introductions: • Siberian • Bearded • Louisiana • Species/Varieties2 |
$35 $75 $50 $25 |
Cost of Production
Production costs will differ based on an operation’s supply sources, but they include all direct costs incurred in production of the final product minus shipping, which is charged by the operation and paid by the consumer. Figure 7 shows an example of how to set up a cost-of-production calculator using a spreadsheet in a program such as Excel (Microsoft, n.d.). These numbers are necessary to inform marketing and sales decisions. In this example, cells are color-coded indicating where operators would input data (number of plants, square footage, cost per plant, etc.), and which cells they would input functions for the spreadsheet’s calculations. Most of the functions are equations that are used to calculate totals. These equations are shown in the white boxes along the right side, with colored arrows indicating which cells they are entered into. A simpler function, which tells the spreadsheet to copy a number from one cell into another, is used in the green color-coded cells (No. 3 in the cell key). An example is noted on the left side of the figure, indicating where a number from cell E8 is automatically input into cell B14. The data for an actual Cost of Production calculator will be an operation’s actual costs, cost estimates and/or other related numbers. This data is entered into cells where indicated.
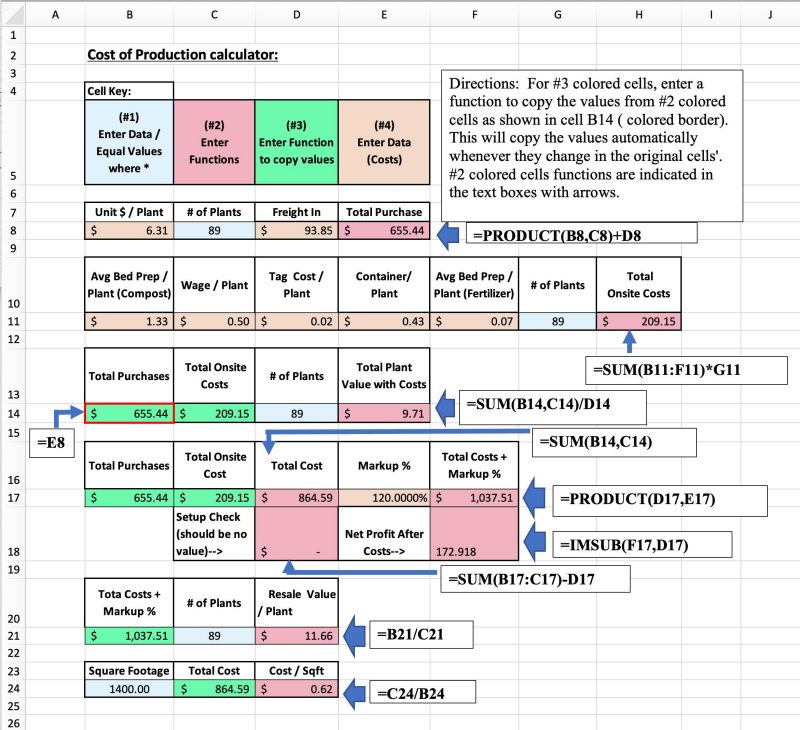
For the purposes of this example, the colors in the cell key (labeled No. 1 through No. 4) help with understanding the setup and what is to be entered where, as data or as a function, to make the calculator work properly, but color-coding is not essential to the setup of the calculator. Excel has a function menu denoted by “fx” in the menu’s bar at the top of the application window, which is an easy way to select what is to be calculated, enter the number ranges, and have the program set up functions in specific cells. Other spreadsheet programs may have a similar menu, and it is not hard to learn how to enter functions following these examples. This example shows how costs figure into determining a resale value and how a markup figures into the calculations to turn a profit. It is not all-inclusive. Each operation’s calculator will reflect its unique costs, but experimenting with this kind of setup using real numbers or estimates can help set sales price goals and clarify the effects of costs and markup on profit, based on the size of the operation.
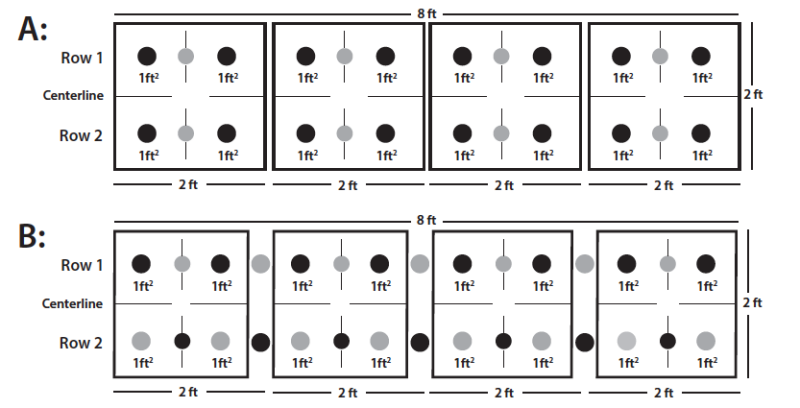
Plant Spacing
Appropriate plant spacing will increase stock and optimize production by getting the greatest number of plants per square foot of production space. Figure 8 shows two different spacing diagrams for iris production. Diagram A works if the plant stock is large and quantity is nearly equal to one plant per square foot of production space. Diagram B works if there are more plants per square foot and the plant stock is smaller. Smaller plant stock can be planted more densely and staggered to establish more plants within the same production space.
The growth patterns of irises are predictable, which allows for determining optimum spacing (figure 9 A and D) and quantities a given area of bed space can support based on their rate of increase. Most irises grow by annual vegetative increase, which may be called fans, that form along the front and sides (figure 9 B and C) outward in a semicircular pattern from the parent plant. Siberian irises are an exception, growing outward generally in a circular pattern away from the original parent plant. Fan divisions, which can be separated as clumps, should occur as needed or on a schedule to maintain optimum growth within a given production space to avoid overcrowding (table 3). Division frequency depends on the type of iris and how vigorously it grows (Schreiner’s, n.d.). For Siberian irises, any older portion of the small rhizomes can be removed and discarded during transplanting, especially if already dead. With Louisiana and bearded irises, the old rhizome, even without any green shoots, may still have new divisions attached or be healthy enough to form new divisions, increasing stock for an extra year. These old rhizomes will eventually die and rot away adding a small amount of nutrients back into the growing media. It is acceptable to discard or replant an older rhizome depending on its condition at the time of transplant. Aside from this one exception, only healthy fan divisions with green shoots and good roots should be transplanted.

| Plant Stock: | Fan Divisions to Transplant as a clump: | Division Interval: |
|---|---|---|
| • Bearded • Louisiana • Siberian |
1-2 1-2 3-4 or more |
3-4 years 3-4 years 4+ years |
Planting Depth
Bearded and beardless irises prefer different planting depths that aid directly in successfully producing them. Rhizomes of bearded irises should be planted in the upper 1-2 inches of the growing media, so the tops of the rhizomes are just barely exposed at the surface along with the crown (SIG, n.d.). Both Siberian and Louisiana irises prefer their rhizomes planted about one inch below the surface of the growing media with an equally deep layer of mulch applied afterward. Replenishing the mulch will benefit beardless irises as it degrades into the growing media (SSI, n.d., ICG, n.d.).
Pest Management
Integrated pest management (IPM) is an assessment-based, environmentally friendly, cost-effective, long-term way to manage pests in an operation. IPM uses a combination of cultural, biological, mechanical, and chemical solutions to pest issues (Blevins, Frank and Beegle 2020, Latimer 2016).
Integrated Pest Management
Many pests can be managed through good bed sanitation. Two examples of IPM techniques include cleaning tools between uses to avoid spreading diseases between beds, and controlling weeds, which compete for light and nutrients and harbor potential insect or disease pests. Another example is to closely monitor irrigation. A drip system keeps foliage dryer than an overhead system. A rain sensor will turn off the irrigation system when there is sufficient rainfall. Dryer plant foliage and well-drained soil prevents disease (Shear 2002). Other IPM strategies include understanding a pest’s lifecycle to know when to intervene with treatments, minimizing pesticide use, using the lowest toxicity products, mulching to moderate soil moisture and control weeds, purchasing pest-free high-quality plant stock, immediately culling out and disposing of sick plants, releasing and or supporting beneficial/predatory insect populations, and frequently scouting to catch and deal with pest problems early (Blevins, Frank, and Beegle 2020, Herbert 2020).
IPM will not necessarily eliminate all pests, but it should keep pests in check at low, tolerable levels (Blevins, Frank, and Beegle 2020). More specific information on IPM can be found in Virginia Tech’s “Home Grounds and Animals” (Virginia Cooperative Extension Publication 456-018) and “Horticultural and Forest Crop Pest Management” (VCE Publication 456-017) guides (Close and Latimer 2020, Day and Hong 2020).
The major steps of an IPM program are (Blevins, Frank, and Beegle 2020):
Set aside time to scout frequently and deliberately.
Use sticky tape, yellow sticky cards, or other traps to monitor pest emergence and presence.
Identify the pest.
Monitor the pest population and assess the damage.
Determine a threshold for when management action is needed (e.g., the point at which the economic loss becomes intolerable).
Examine cultural, biological or mechanical management options that also include taking no action, as well as chemical treatment options.
Choose the least toxic alternative combination of management options that reduces the economic threshold.
Evaluate how well the IPM techniques worked.
Create a pest management journal or database that includes pictures, notes, solutions, evaluations, and considerations for future management decisions.
Try to prevent the pest problem from occurring again.
Insects
Become familiar with the common pests of the crop. The main insect pests in iris are aphids (figure 10) and the iris borer (figure 11). Aphids can be washed or brushed off the plants or managed with predatory beneficial insects or insecticides (Day 2020). Damage from aphids is generally cosmetic, but they can carry diseases. The iris borer is a native moth larva which is found mainly in the Eastern U.S. (Shear 2002, Dellinger and Day 2015). The borers attack both native irises and nonnative bearded iris and hybrids, but they seem to do more damage to nonnatives. The borer life cycle (figure 11) is easily interrupted with annual debris cleanup and immediate removal of damaged plants (Shear 2002, Dellinger and Day 2015).


Diseases
Iris diseases are caused mainly by fungi or bacteria (table 4). Viruses and conditions with unknown origins, such as scorch, account for a very small percentage of iris disease occurrences. With viruses, affected plants are removed. With scorch, the condition is generally not fatal or contagious, but plants either recover with basic treatment like cutting off affected foliage, or they succumb without infecting other plants nearby (Shear 2002, Warburton and Hamblen 1995).
| Pathogen | Symptoms | Conducive Conditions | Treatment |
|---|---|---|---|
| Bacterial Leaf Blight (Bacterial leaf spot) (Xanthomonas tardicrescens) | Similar to fungal leaf spots. Spots can run together forming nondistinct smears. Heavy exudate as the disease progresses. Presents as tiny, watery pinholes and thin short track marks no wider than a ballpoint pen tip at leaf margins. | Wet weather and mild temperatures. | Good bed and tool sanitation, 6% bleach solution to dip tools between uses. Disposable gloves when working in infected areas. Cleanups can remove inoculum before it ever causes symptoms. |
| Fungal Leaf Spot (Didymellina macrospora) Other Pathogens: Alternaria iridicola, Ascochyta iridis, Asteroma venulosus, Cylindrosporium iridis (most common), Mycosphaerella macrospore, etc. | Brown circles on leaf tips with dark centers and spores. May briefly have a watery appearance as they mature. | Wet, humid and windy conditions, as well as careless hygiene practices. Working with infected plants under wet, humid, windy conditions. | Regular removal of old leaves and infected leaves. Keep leaves dry. Increase space between plants for air circulation. Fungicides can be helpful. |
| Bacterial Soft Rot (Erwinia carotovora) | Rotting rhizomes smell like rotten potatoes. Leaf fans wilt, die, and can be pulled free from the rhizome at the crown. In advanced stages, rhizomes are yellowish and mushy. | Wet weather and mild temperatures. Becomes inactive during the winter months. | Remove affected tissue. Expose cleaned rhizomes to sun, allow to dry, replant. Avoid wounding rhizomes. Control insects that feed on rhizomes. Disinfect tools between each use. Dispose of diseased material in trash. No known chemical treatment. |
| Fungal Soft Rot (Crown Rot) Sclerotium rolfsii-Mustard Seed Fungus | Yellowing leaf tips with basal rot and leaf fan collapse. Rhizome rot by secondary infections of bacterial soft rot. Gray or tan cotton-like masses on adjacent soil, rhizomes and leaf bases. | Warm weather with any moist soil. | Bed sanitation and cleanups. Exposure of plants to sun once cleaned of infected tissue. For advanced infections, dig plant completely, remove infected tissue, and treat with sun, replanting in new soil and location. |
| Fungal Soft Rot (Botrytis Rot) (Sclerotinia (Botrytis) convoluta - Botrytis Rhizome Rot) | Black mold over the outer surface of infected rhizomes and roots. Plants do not grow. Rhizomes are brown instead of white inside. | Cool wet weather. In fall, rhizomes suffer from dry rot. New growth does not appear the following spring. | None; rhizomes must be cleaned of infected material, dried, and replanted in clean soil to stop the disease if present. |
| Iris Rust (Puccinia iridis) (very rare) | Small rusty-brown lesions over entire leaf surface. Dusty spores as with all rusts. Stunted growth. | High humidity and moderate temperatures. Excess moisture. Fogs, dews, rain, or irrigation systems encourage spread and growth. | Sanitation and careful cultivar selection to grow resistant plants. Careful removal of infected tissue or whole plants is effective. Irrigate beds when plants can dry out prior to nightfall. |
| Scorch (unknown pathogen) | Infected rhizomes remain firm while roots rot and die. Foliage exhibits a dwarfing effect with lack of growth and yellowing of central leaves. Leaves cannot be pulled free of crown. | Unknown | Unknown, but some plants have been successfully treated by digging infected rhizomes, drying in the sun for a few days to weeks, then replanting. |
| Pineappling (unknown pathogen if any) | Enlarged rhizomes. Dwarfed foliage folded or pleated. Flower stems stunted, clubbed and malformed. Underdeveloped roots. | Unknown. Occurs sporadically but does not appear transmissible between plants. | Unknown. Affected plants either die or resolve the condition on their own. |
| Bearded iris mosaic virus, Iris mild mosaic virus, and Iris severe mosaic virus. | Reduced vigor and quality, stunting, light green stripes or streaks, teardrop-shaped spots on leaves, mottling of flower stalk. | Spread by aphids or other sucking insects. | Control aphids. Cull out and dispose of plants in the trash. Quarantine plants to see if virus appears next season. |
Business Management
Business management includes many different aspects of an operation. When starting and/or running a small scale operation, the owner may perform most of the tasks. While each business is unique, some considerations are common to them all.
Business Plan and Budget
Creating a business plan is a good way to organize information, develop a budget, and think through what is needed, when it’s needed, and how to obtain it. Resources for creating business plans are available from small business associations and various online sources. A well-done business plan and budget have many uses including (White 2007):
Identifying all of the inputs needed to produce the business.
Identifying the top five expenses for cost control management.
Determining potential changes in the business.
Estimating the minimum operating loan that might be requested from a lender.
Determining how much revenue can be generated from the business.
Determining a break-even analysis for price and yield.
Software
Select software that can best accommodate business needs. Commonly available software packages can save time and money, are usually user friendly, and have technical support available. Customized software and outsourcing tasks can be considered as the budget allows. Tasks that normally require software include:
Communications (email, website, social media, publicity, and marketing).
Office activities (word processing, spreadsheets, photo/design/presentation, and marketing materials).
Data management (plant and hybridizing records, cultural information, and checklists).
Inventory management (availability, sales, stock, pricing, supplies, re-orders, and shipping/tracking).
Record-keeping (billing/order fulfillment, sales, taxes, payroll, purchases, production costs, revenues, operating expenses/variable costs); financial analysis/evaluation (quarterly and annual reports, cost analysis, long-term goals and benchmarks, progress reports, trends, problem identification, revenues vs. operating expenses, fixed costs, and assets).
Marketing
Marketing covers a broad spectrum of items. Everything from how the operation presents itself physically to its advertising campaigns will involve some aspect of communication with prospective customers. Choices about marketing should be reasoned and informed, especially when dealing with a limited budget, in order to be effective. Some things may not work even when done well, but it is important to understand what does not work and why in order to find what suits the operation.
While the sale of bare-root plants is the prime focus for small scale iris operations, both the cut flower and potted plant markets should be given serious consideration. Onsite sales of stems or working with local florists may yield untapped revenue streams and market exposure while diversifying a product line with little added effort and expense. Running a booth at a local farmers or flea market can be an outlet for potted irises sourced from the old rhizomes normally discarded during annual replantings of those cultivars that require division; turning a waste stream into a sellable product. In addition, stock that is being phased out due to operational considerations may still be marketable to local garden centers as potted perennials. Selling off unwanted stock that is still commercially viable can help support production of newer cultivars.
The following questions are important in any marketing approach:
What exactly is being marketed?
Who is the target market audience?
How do they receive their information?
What are the best marketing outlets/platforms/avenues for the product?
What marketing materials need to be developed (logo, business cards, content for website and social media)?
How will customers be engaged and responded to?
How will plants be delivered or shipped? Mail order only, local delivery using the operation’s own vehicle or common carrier, pickup at nursery?
Will e-commerce be used to reduce face-to-face contact and/or streamline sales?
Are there geographic marketing boundaries that need to be addressed with permits and inspections (local, regional, national, international) such as for the U.S. Department of Agriculture Animal and Plant Health Inspection Services (APHIS, n.d., VDACS, n.d.)?
Are there domestic regulations that impact marketing (inspections/certificates)? Each state has a nursery permit requirement for interstate commerce such as the Virginia Department of Agriculture and Consumer Services (VDACS, n.d.). What types of permits are required for selling only cut flower stalks and potted plants within the state or a combination of both in interstate commerce?
Summary
Starting a small scale perennial production operation can be challenging and exciting, and with sound decisions, it can be done well and in a sustainable and profitable manner. While there is no guarantee, each nursery operation has its own strengths which, if built upon, can aid in its success. Getting to know neighbors by visiting other nurseries in the area will help build an important support network where operators can learn effective solutions to commonly encountered challenges. In the end, compliance with formal regulations and ordinances for an operation’s location will still leave much room for the creativity and ingenuity that comes from addressing one’s own specific challenges in setting up and running a new operation to ultimately make it self-sufficient.
Additional Resources
1001pallets. n.d. Accessed September 17, 2020. https://www.1001pallets.com/pallet-safety/.
American Daylily Society. n.d. “Daylily Dictionary.” Accessed November 14, 2020. https://daylilies.org/daylily-dictionary/.
The American Iris Society (AIS). n.d. Main page. Accessed November 14, 2020. https://www.irises.org/.
AmericanHort. 2014. “The American Standard for Nursery Stock,” ANSI Z60.1. Columbus: American Horticulture Industry Association.
Beal, B., G. Bowen., P. Goeringer, and M. Todd. 2019. “Considerations for Acquiring a Farm: Selecting the Best Farm Property,” Publication FS-1094. University of Maryland Extension, College Park. https://extension.umd.edu/resource/considerations-acquiring-farm-selecting-best-farm-property.
Easton, M. Z. and E. Bock. 2016. “Soil and Soil Water Relationships,” VCE Publication BSE-194P. Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/BSE/BSE-194/BSE-194.html.
Eaton, G. K., B. Appleton, and J. Owen. 2014. “Getting Started in the Nursery Business: Nursery Production Options,” VCE Publication 430-050. Virginia Cooperative Extension, Blacksburg. Download available at https://vtechworks.lib.vt.edu/bitstream/handle/10919/48106/430-050_pdf.pdf.
Fox, L. J., D. J. Robinson., D. J. Sample, and G. E. Nelson. 2018. “Stormwater Management for Homeowners Fact Sheet 2: Rain Barrels,” VCE Publication SPES-10P. Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/SPES/SPES-10/SPES-10.html.
Freeborn, J., and D. Relf. 2018. “Landscaping for Less in the Landfill,” VCE Publication 426-716 (HORT-243NP). Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/426/426-716/426-716.html.
Groover, D., and D. Osborne. 2017. “Farm Record Book: Expenses and Receipts,” VCE Publication 446-017 (VCE-867NP). Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/446/446-017/446-017.html.
Haering, K. C., G. K. Evanylo, B. Benham, and M. Goatley. 2018. “Water Reuse: Using Reclaimed Water for Irrigation,” VCE Publication 452-014 (SPES-1P). Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/452/452-014/452-014.html.
Hunnings, J. R., S. J. Donohue., and S. Heckendorn. 2019. “Soil Sampling for the Home Gardener,” VCE Publication 452-129 (SPES-176P). Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/452/452-129/452-129.html.
Latimer, J., and J. Owen. 2016. “Resources for Greenhouse and Nursey Operations and Operators,” VCE Publication 430-104 (HORT-188P). Virginia Cooperative Extension, Blacksburg. https://resources.ext.vt.edu/contentdetail?contentid=3027.
Ling, E., B. Benham. 2019. “Rainwater Harvesting Systems,” VCE Publication BSE-116NP (BSE-265NP). Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/content/pubs_ext_vt_edu/en/BSE/BSE-116/BSE-116.html.
Maguire, R., and S. Heckendorn. 2019. “Soil Sampling Instructions for the Farm,” VCE Publication SPES-141NP. Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/content/pubs_ext_vt_edu/en/SPES/SPES-141/SPES-141.html.
Mountain, T., and M. A. Walker. 2018. “Virginia Retail Sales and Use Regulations,” VCE Publication AAEC-159NP. Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/AAEC/AAEC-159/AAEC-159.html.
Nartea, T., and K. L. Morgan. 2015. “Selling Directly to Buyer: How to Price Your Products,” VCE Publication AAEC-97P. Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/content/dam/pubs_ext_vt_edu/AAEC/AAEC-97/AAEC-97-PDF.pdf.
Niemiera, A. X, 2018. “Diagnosing Plant Problems,” VCE Publication 426-714. Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/426/426-714/426-714.html.
Niewolny, K., C. Kloetzli, and A. Vallotton. 2013. “Successful Farm Startup for Beginners: Virginia Beginning Farmer and Rancher Coalition Program,” VCE Publication AEE-67NP. Virginia Cooperative Extension, Blacksburg. https://resources.ext.vt.edu/contentdetail?contentid=2286.
Niewolny, K., A. Mark., K. Morgan., P. Callan., K. Scott., T. Nartea, and J. Hilleary. 2020. “Introduction to Labor Issues for Beginning Farmers,” VCE Publication ALCE-106NP (ALCE-186NP). Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/content/dam/pubs_ext_vt_edu/AEE/AEE-106/ALCE-186.pdf.
Prince William County Service Authority (PWCSA). n.d. “Sample Bill.” Accessed September 19, 2020. https://www.pwcsa.org/sample-bill.
Relf, D. 2020. “Vegetable Gardening in Containers,” VCE Publication 426-336 (SPES-255P). Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/426/426-336/426-336.html.
Relf, D., and E. Ball. 2009. “Perennials: Culture, Maintenance and Propagation,” VCE Publication 426-203. Virginia Cooperative Extension, Blacksburg. http://hdl.handle.net/10919/48235.
Scoggins, H. 2019. “Getting Started in the Production of Field-Grown, Specialty Cut Flowers,” VCE Publication 426-618 (SPES-171P). Virginia Cooperative Extension, Blacksburg. https://www.pubs.ext.vt.edu/426/426-618/426-618.html.
Society for Louisiana Irises. n.d. Main page. Accessed November 14, 2020. http://www.louisianas.org/.
Society for Siberian Irises. n.d. Main page. Accessed November 14, 2020. http://www.siberianirises.org/.
Stehouwer, R. 2002. “Environmental Soil Issues: Garden Use of Treated Lumber,” Publication CATUC173. The Pennsylvania State University, University Park. https://extension.psu.edu/environmental-soil-issues-garden-use-of-treated-lumber.
University of California Agricultural and Natural Resources: Statewide Integrated Pest Management Program. n.d. “Pest in Gardens and Landscapes.” Accessed October 22, 2020. http://ipm.ucanr.edu/PMG/GARDEN/FLOWERS/iris.html.
Glossary
bare root – Harvested plants from which the soil or growing medium has been removed.
bearded irises – Any of numerous wild or cultivated irises with a growth of short hair-like structures on each fall.
beardless irises – Any of numerous wild or cultivated irises having no growth of short hairs on the falls.
clump – Specific to daylilies, this term refers to three or more fans of a cultivar grouped together. A more general usage, applied to irises, simply refers to any grouping of fans separated from the larger grouping during division of a larger grouping that can be effectively transplanted to grow new plants and increase the plant stock.
corolla – A collective term for the petals of a flower.
crown – The total of an individual plant’s aboveground parts, including stems, leaves, and reproductive structures. In irises, it is generally referred to as the growing point of the plant at the soil’s surface from which the aboveground parts develop.
cultivar/s – Short for cultivated variety. A plant variety that has been either produced naturally and selected by man or produced by man in cultivation through selective breeding. Cultivars do not reproduce true to seed, so must be asexually propagated. The first letter of a cultivar is capitalized and the name is not italicized. Cultivars are either surrounded by single quotation marks or preceded by the abbreviation “cv.”
cultivator – A mechanical implement for breaking up the soil and uprooting weeds. A cultivator is primarily used to mix loose soil. It is unlikely to work to create a new growing space due to its light-duty tines.
division/s – The action of separating something into parts or the process of being separated. In horticulture and gardening, is a method of asexual plant propagation, where the plant (usually an herbaceous perennial) is broken up into two or more parts. Both the root and crown of each part is kept intact. Also, a term for describing an individual unit of a clump, containing leaves, crown, and roots identical to the parent plant. Interchangeable with the terms “fan” and “ramet.”
emitter/s – A device used to distribute water for irrigation that can discharge in droplets, small streams, or through mini-sprayers.
falls – The three lower petals of the iris flower, which are modified sepals, that may either hang down, flare out, or curl under the bloom.
fan – Descriptive term for the growth habit of certain perennial plants, such as irises and daylilies, which have no vertical stem because the leaves originate from the rhizome. May precede the term “division” to provide a more descriptive compound term for an individual unit of a clump, containing leaves, crown, and roots identical to the parent plant. The term is interchangeable with the terms “division” and “ramet.”
growing media – Any substance through which plant roots grow and extract water and nutrients. It can act both as a reservoir and/or source for nutrients depending on its composition. Both mineral soils, which provide nutrients directly, and peatmoss-based planting media, which do not, are examples of growing media.
historic irises – Irises that have been actively sold in commerce for at least 30 years attain this designation. They can still be sellable in a more specialized market for such older irises.
increase – A general shortening of the term “vegetative increase.”
mulch – Any material that is left on the soil surface. Its purpose is to reduce evaporation and runoff, inhibit weed growth, and create an attractive appearance. Mulches also moderate soil temperature. Organic mulches may be incorporated into the soil as amendments after they have decomposed to the point that they no longer serve their purpose.
NOID – A designation specific to irises as a shorthand version of “No Identification” (abbreviated as NOID) to indicate any plant unidentifiable by registered name.
nutrient – A substance that provides nourishment essential for plant growth and the maintenance of life. It differs from a soil amendment in that it does not improve soil’s physical properties.
ornamental crops – Plants that are grown for decorative purposes in gardens and landscape design projects.
pesticide – A chemical substance used for managing insects or other organisms harmful to cultivated plants or to animals.
petals – Structures collectively making the corolla, which protect the inner reproductive structures of a flower and often attract insects by either their color or their nectar and thus facilitate pollination.
plant stock – Any plant or plant tissue, including rhizomes, shoots, leaf or stem cuttings, roots, crowns, or tubers, used in plant production or propagation.
ramet – Individual plants in a clump, each portion of which is identical with the original parent plant. Interchangeable with the terms “fan” and “division.”
rate of increase – A measure of the vigor of a cultivar, specifically irises, referring to how many new iris plants are produced within a defined time period. Dividing the total number of plants of a given cultivar by the number of years over which it has been grown gives the annual rate of increase. Other rates might be a per clump rate of increase where you divide the same total number of plants by the number of clumps. For irises, rates of increase greater than three are preferred in order to generate sufficient production quantities for commercial sales.
rhizome/s – A continuously growing horizontal underground stem that puts out lateral shoots and adventitious roots at intervals. Also described as the underground portion of stem modified for energy storage by plants such as irises, which supports roots and the vegetative potions of the plant.
sepal – In a flower, one of the segments or divisions of the outer whorl of nonfertile parts surrounding the fertile organs; usually green.
soil amendments – Any material added to a soil to improve its physical properties, such as water retention, permeability, water infiltration, drainage, aeration, and structure. The goal is to provide a better environment for roots.
standards – The three upright petals of the iris flower.
tiller – A mechanical implement designed to break up hard, compacted soil into loose, smaller particles that are better for planting.
variety/varieties – A naturally genetically different plant or group of plants within a species that has one or more distinguishing characteristics and usually produces true to seed.
vegetative increase – The new plant developing along the side of the parent plant annually at or just below the soil surface, specifically used when referring to irises and their annual growth cycle. Many times, simply referred to as “increase” with the plural being “increases.”
whorl – A ring of organs borne at the same level on an axis (e.g., leaves, bracts, or floral parts).
Acknowledgements
The authors would like to express appreciation for the reviews and comments provided by the following individuals: Mike Andruczyk, Horticulture Agent, Virginia Cooperative Extension City of Chesapeake Office; Chris Mullins, Virginia Cooperative Extension Specialist, Virginia State University, Greenhouse and Specialty Crops Program; Anthony V. LeBude, Associate Professor, Nursery Crops, North Carolina State University Extension and Research; Susan Cheek, Agriculture Management Agent, Virginia State University, Small Farm Outreach Program. Further acknowledgements for support, feedback, contributions and insight for this publication go to: Rick Tasco and the late Roger Duncan of Superstition Gardens; Don and Ginny Spoon of Winterberry Iris Gardens; The American Iris Society; Society for Siberian Irises; Society for Louisiana Irises; The Fredericksburg Area Iris Society; American Hemerocallis Society; and Douglas and Joyce Eagles.
References
AIS (The American Iris Society). 2011. Basic Iris Culture: What Every Iris Grower Should Know, 3rd ed.
AIS (The American Iris Society). 2018. “Directory Listing.” Accessed October 14, 2020. https://www.irises.org/resources/commercial-directory/.
APHIS (U.S. Department of Agriculture Animal and Plant Health Inspection Service). n.d. Home page. Accessed November 18, 2020. https://www.aphis.usda.gov/aphis/home/.
Blevins, S., D. Frank, and D. Beegle. 2020. “An Introduction to Integrated Pest Management,” VCE Publication ENTO-365NP. Virginia Cooperative Extension, Blacksburg.
BHIG (Bluebird Haven Iris Gardens). n.d. Online catalog. Accessed November 14, 2020. http://www.bluebirdhavenirisgarden.com/siteV2/catalog.html.
Close, D., and J. Latimer. 2020. “Pest Management Guide: Home Grounds and Animals,” VCE Publication 456-018. Virginia Cooperative Extension, Blacksburg.
Day, E. R. 2020. “Aphids,” VCE Publication 444-220. Virginia Cooperative Extension, Blacksburg.
Day, E. R., and C. Hong. 2020. “Pest Management Guide: Horticultural and Forest Crops,”
VCE Publication 456-017. Virginia Cooperative Extension, Blacksburg.
Decadent Daylilies. n.d. “Louisiana Irises for Sale.” Accessed October 6, 2020. https://www.decadentdaylilies.com/shop/louisiana-iris/.
Dellinger, T. A., and E. R. Day. 2015. “Iris Borer,” VCE Publication ENTO-140NP. Virginia Cooperative Extension, Blacksburg.
Dripdepot. n.d. “Irrigation System Design Help.” Accessed November 14, 2020. https://www.dripdepot.com/help/irrigation-system-design-howto.
Dripworks. n.d. “Gallery of Plans.” Accessed November 14, 2020. https://www.dripworks.com/gallery-of-plans.
Freeborn, J., and D. Relf. 2017. “Building Healthy Soil,” VCE Publication 426-711. Virginia Cooperative Extension, Blacksburg.
Hahn, J., and J. Fetzer. 2018. “Iris Borers.” University of Minnesota Extension, Saint Paul. https://extension.umn.edu/yard-and-garden-insects/iris-borers.
Hahn, J., and S. Wold-Burkness. 2019. “Aphids in Home Yards and Gardens.” University of Minnesota Extension, Saint Paul. https://extension.umn.edu/yard-and-garden-insects/aphids.
Hansen, M. A. 2016. “Iris Leaf Spot,” VCE Publication 450-600. Virginia Cooperative Extension, Blacksburg.
Herbert, D.A. 2020. “Integrated Pest Management Peanut Scouting Manual,” VCE Publication 444-126. Virginia Cooperative Extension, Blacksburg.
Hillcrest Irises. n.d. “Shop.” Accessed October 6, 2020. https://www.hillcrestiris.com/shop/.
HIPSa (Historic Iris Preservation Society). n.d. “Commercial Sources.” Accessed October 22, 2020. https://historiciris.org/resources/historic-iris-marketplace/.
HIPSb (Historic Iris Preservation Society). n.d. “Iris Glossary”. Accessed October 22, 2020. https://historiciris.org/hips-library/iris-glossary/.
ICG (Iris City Gardens). n.d. “Growing Tips.” Accessed October 3, 2020. http://www.iriscitygardens.com/frame1.html.
Joe Pye Weed’s Gardens. 2020. Online catalog. Accessed October 6, 2020. http://www.jpwflowers.com/.
Kohls, R. L., and J. N. Uhl. 2015. Marketing of Agricultural Products. Pradesh: Pearson India Educational Services.
Latimer, J. 2016. “Applying Pesticides Safely,” VCE Publication 426-710. Virginia Cooperative Extension, Blacksburg.
Microsoft. n.d. “Microsoft Excel.” Accessed October 6, 2020. https://www.microsoft.com/en-us/microsoft-365/excel.
Mister Landscaper. n.d. “How to Get Started.” Accessed October 1, 2020. https://misterlandscaper.com/how-to-get-started.
Moorman, G.W. 2016. “Iris Diseases.” Pennsylvania State University Extension. https://extension.psu.edu/iris-diseases.
Niemiera, A. X. 2018. “Intensive Gardening Methods,” VCE Publication 426-335. Virginia Cooperative Extension, Blacksburg.
Orbit. n.d. “Sprinkler System Designer.” Accessed October 1, 2020. https://www.design.orbitonline.com/.
PWSWCD (Prince William County Soil and Water Conservation District). n.d. “Traditional Agriculture.” Accessed November 14, 2020. https://www.pwswcd.org/traditional-agriculture.
Rainbird. n.d. “Design Resources.” Accessed October 1, 2020. https://www.rainbird.com/professionals/specifier/specifierhome.
Schreiner’s Iris Gardens. n.d. “How to Grow and Care for Iris.” Accessed October 3, 2020. https://www.schreinersgardens.com/.
Shear, William. 2002. The Gardener’s Iris Book. Newtown: The Taunton Press.
SIG (Sutton’s Iris Gardens). n.d. “Iris Care.” Accessed November 15, 2020. https://www.suttoniris.com/pages/iris-care.
SIGNA (Species Iris Group of North America). n.d. Accessed October 22, 2020. http://www.signa.org/.
SNA (Southern Nursery Association). 2013. Best Management Practices-Irrigation. Accessed August 30, 2020. https://ellisonchair.tamu.edu/2013/12/29/best-management-practices-guide-for-producing-nursery-crops/.
SSI (Society for Siberian Irises). n.d. “Siberian Iris Culture.” Accessed November 15, 2020. http://www.siberianirises.org/ssicult.htm.
Straw, R. A. 2015. “Irrigating the Home Garden,” VCE Publication 426-322. Virginia Cooperative Extension, Blacksburg.
Trinklein, D. H. 2014. “Raised-Bed Gardening,” Publication g6985. University of Missouri Extension, Columbia.
USPTO (The United States Patent and Trademark Office). n.d. “Search for Patents.” Accessed February 6, 2021. https://www.uspto.gov/patents/search.
Uva, W. L. 2009. “Smart Pricing Strategies,” VCE Publication 2906-1324. Virginia Cooperative Extension, Blacksburg.
VDACS (Virginia Department of Agriculture and Consumer Services). n.d. “Nursery Inspection.” Accessed November 18, 2020. https://www.vdacs.virginia.gov/plant-industry-services-nursery-inspection.shtml.
Warburton, B., and M. Hamblen, editors. 1995. The World of Irises, 3rd ed., 340-342. Salt Lake City: The Publisher’s Press.
Weil, R. R., and N. C. Brady. The Nature and Properties of Soils, 5th ed. New York: Pearson, 2017.
White, A. 2007. “Financial Analysis of an Agricultural Business — the Enterprise Budget.” Virginia Cooperative Extension Newsletter Archive, Virginia Cooperative Extension, Blacksburg. Accessed October 6, 2020. https://www.sites.ext.vt.edu/newsletter-archive/fmu/2007-04/financial_analysis.html.
Winterberry Iris Gardens. 2011. “Tall Bearded Iris Prices.” Accessed October 6, 2020. http://www.winterberryirises.com/tall_bearded_irises_prices.
Virginia Cooperative Extension materials are available for public use, reprint, or citation without further permission, provided the use includes credit to the author and to Virginia Cooperative Extension, Virginia Tech, and Virginia State University.
Virginia Cooperative Extension is a partnership of Virginia Tech, Virginia State University, the U.S. Department of Agriculture (USDA), and local governments, and is an equal opportunity employer. For the full non-discrimination statement, please visit ext.vt.edu/accessibility.
Publication Date
April 20, 2022



