Introduction to Environmental Monitoring Programs
ID
FST-290NP
Overview
Environmental monitoring programs (EMPs) are a proactive approach that operations may take to reduce microbial contamination events. EMPs are designed to assess the effectiveness of the hygiene and sanitation practices in a food environment and provide valuable information to avoid microbial contamination of food products. EMPs are specific to each operation and depend upon the food(s) that are handled, packed, and or processed; operation size; operation layout; among other factors. If EMPs are effective, operations will be alerted to possible pathogen harborage locations based on positive test results, and operations will be able to implement corrective actions to reduce contamination events.
Corrective actions may include targeted cleaning and sanitation of locations/adjacent locations that test positive and implementation of new policies or practices to reduce cross-contamination (for example, new employee or equipment traffic patterns).
Assemble the Team and Identify Zones
The first step to creating a successful EMP is to assemble a knowledgeable team. Possible members of an EMP team include a microbiologist, line supervisor, operations manager/staff, quality manager/staff, sanitation manager/staff, and or maintenance manager/staff. As part of developing an EMP, the team must divide the food operation (for example, packinghouse, processing facility) into four sanitary zones from highest risk areas (Zone 1) to lowest risk areas (Zone 4) of contamination. Examples below:
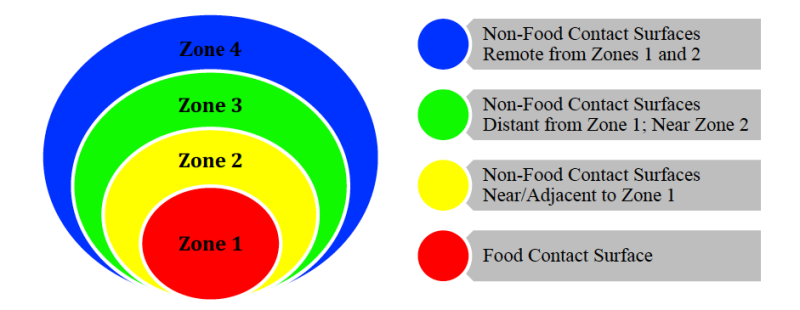
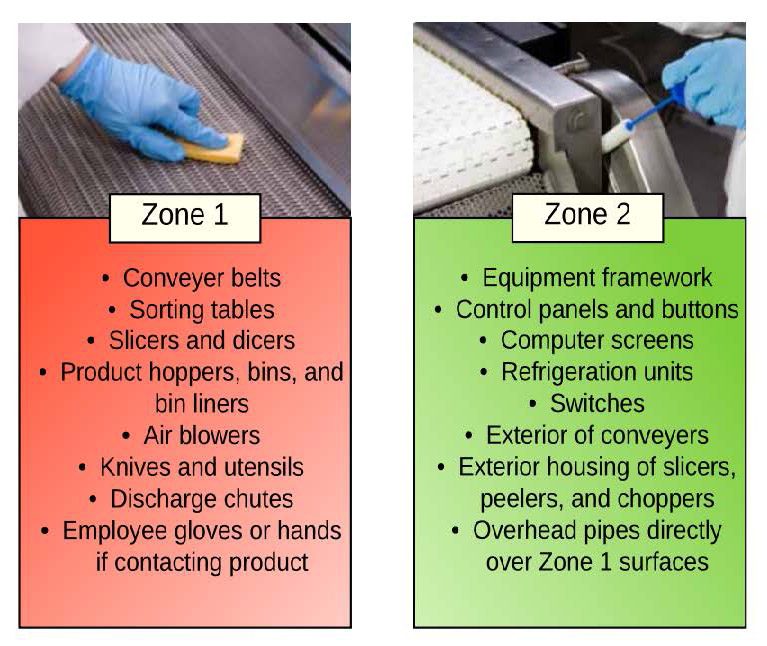
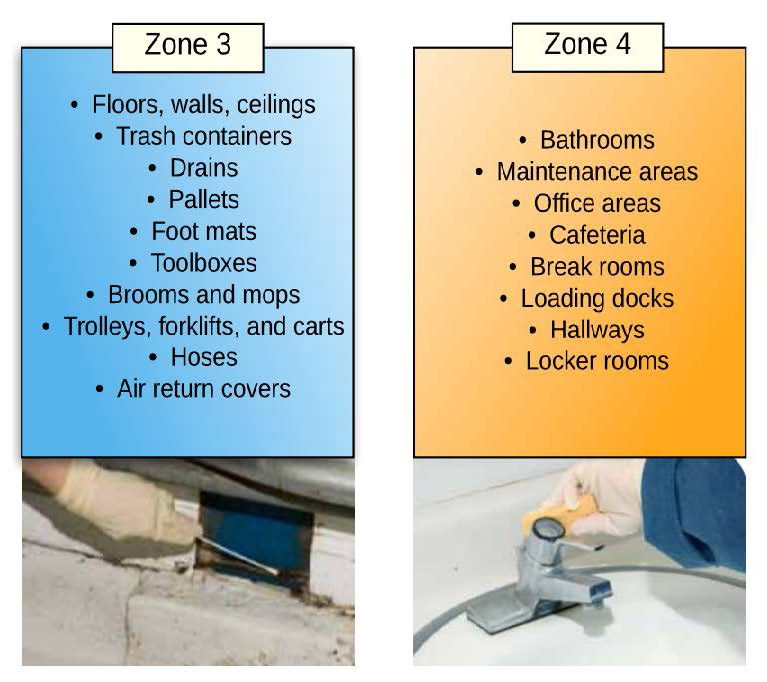
Images Adapted from the document: Pathogen Environmental Monitoring Program created by the Almond Board of California
Selecting Locations to Sample
EMPs can help operations identify vulnerable areas (in other words areas where contamination may be more likely to occur) in an operation (for example, packinghouse or processing environment). When selecting locations to sample in operations, it is recommended to focus on locations that are funnel points or high traffic flow areas. For example, take samples where produce builds up like at the end of the packing line (a funnel point). Often operations will collect samples during peak operation (3-4 h into the shift) to capture the worst-case scenario (as this sampling point would represent typical operating conditions; and may also represent times when produce is most prone to contamination). If operations wish to verify sanitation programs (cleaning and sanitizing of equipment, tools, and buildings), EMP samples may be collected post-sanitation.
Microbiological Testing
EMPs often involve testing for indicator organisms in these four zones. Indicator organisms are non- pathogenic microorganisms that are more numerous than specific pathogenic microorganisms (making them easier to find) and often sharing similar characteristics to pathogens. For example, Listeria species are often used as an indicator for the pathogen Listeria monocytogenes. Typically, if an indicator organism is found in a location upon testing, the theory is a pathogen could also be found in that location. Indicator organisms are also good to use because they are commonly found in higher numbers than pathogens; therefore, an operation may identify an operational weakness before a contamination event occurs. Common indicator organisms are total aerobic bacteria, coliforms, generic Escherichia coli, and Listeria species.
NOTE:
Make sure to establish corrective actions for positive samples in advance!
If and when positive test results are found for indicator organisms, the EMP should outline corrective actions that the operation/team should take to eliminate the source of contamination. The corrective actions should be discussed prior to sampling locations and
microbiological testing; and depend on the zone where the positive result is found and if the positive result is a first-time event or a repeated occurrence. All positive test results should trigger a corrective action (from minor to major actions). Zone 1 (food contact surfaces) positives or repeated positives at a sample location should be handled with most caution (as these types of positives indicate a problem and potential for product contamination). Examples of corrective actions include clean and sanitize location then re-sample; determine root source by concentrated sampling in that area and surrounding areas (vector swabbing); retrain employees; repair or replace equipment; among others (contact a Virginia Cooperative Extension agent/specialist for more information on EMPs and corrective actions). Finally, an effective EMP involves repeated microbiological testing regardless of positive or negative results to assist in continued prevention-based food safety efforts.
References
United Fresh. 2013. Guidance on Environmental Monitoring and Control of Listeria for the Fresh Produce Industry.
http://www2.unitedfresh.org/forms/store/ProductFormPublic/search?action=1&Product_productNumber=42425
Almond Board of California. 2010. Pathogen Environmental Monitoring Programs.
http://www.almonds.com/sites/default/files/content/attachments/pem_book.pdf
T. Freier. 2014. Components in a Pathogen Environmental Monitoring Program.
http://www.foodqualityandsafety.com/article/components-for-an-effective-pathogen-environmental-monitoring-program/
Virginia Cooperative Extension materials are available for public use, reprint, or citation without further permission, provided the use includes credit to the author and to Virginia Cooperative Extension, Virginia Tech, and Virginia State University.
Virginia Cooperative Extension is a partnership of Virginia Tech, Virginia State University, the U.S. Department of Agriculture (USDA), and local governments, and is an equal opportunity employer. For the full non-discrimination statement, please visit ext.vt.edu/accessibility.
Publication Date
November 2, 2023



