Virginia Household Water Quality Program: Corrosive Household Water
ID
442-665 (BSE-259P)
Private water sources such as wells and springs are not regulated by the U.S. Environmental Protection Agency (EPA). Although private well construction regulations exist in Virginia, private water supply owners are responsible for providing maintenance for their water systems, monitoring water quality, and taking the appropriate steps to address problems, should they arise.
The EPA public drinking water standards are good guidelines for assessing your water quality. “Primary drinking water standards” apply to contaminants that can adversely affect health and are legally enforceable for public water systems. “Secondary drinking water standards” are nonregulatory guidelines for contaminants that may cause nuisance problems such as bad taste, foul odor, or staining.
Testing your water annually and routinely inspecting and maintaining your water supply system will help keep your water safe. For more information, visit the Virginia Household Water Quality Program website at www.wellwater.bse.vt.edu.
Introduction
Corrosive water can react with minerals and metals that it comes in contact with to “corrode” or dissolve them. Corrosive water is often called “aggressive” water. While consuming corrosive or aggressive water is not in itself dangerous, consuming some of the contaminants that may be dissolved by corrosive water may pose health risks, particularly metals like copper and lead.
Many homes have pipes and plumbing fixtures that contain copper, lead, or other metals. Given the right conditions, corrosive water can dissolve enough of these metals into water used for drinking and cooking to cause health problems, nuisance effects, or both. There are also conditions under which corrosive water can carry metals from soils, geologic formations, or other sources into the home.
Consuming lead can damage the body’s neurological, cardiovascular, reproductive, and renal systems. Ingesting copper can cause nausea, vomiting, diarrhea, and stomach cramps. The U.S. Environmental Protection Agency (EPA) has set the “maximum contaminant level goal” (MCLG) for lead in drinking water at zero micrograms per liter (0 µg/L), with a health action level of 15 µg/L. The EPA’s MCLG and health action level for copper is 1.3 milligrams per liter (mg/L). In addition, the EPA has established a nuisance or “secondary maximum contaminant level” (SMCL) for copper at 1 mg/L.
An example of a nuisance problem that corrosive water can produce is the blue-green staining on sinks, tubs, or laundry caused by dissolved copper. If left untreated, corrosive water will reduce the life expectancy of water-using appliances like water heaters, washing machines, and dishwashers.
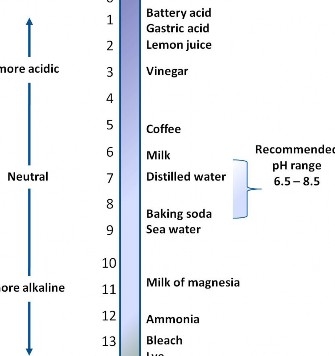
While several factors contribute to the corrosivity of your water, the primary determining factor is the water’s pH. Water pH indicates intensity of water in terms of alkalinity or acidity and values range from zero (most acidic) to 14.0 (most alkaline), with 7.0 being neutral. Figure 1 shows the pH of some common substances. While substances at either end of the pH scale can be corrosive, in private water systems low pH values (less than 6.5) are more common than high values (more than 8.5).
It is important to note that the pH scale is logarithmic. This means that water that has a pH of 6.0 is 10 times more acidic than water with a pH of 7.0, and that water with a pH of 5.0 is 100 times more acidic than water with a pH of 7.0. Small changes in pH can make a big difference.
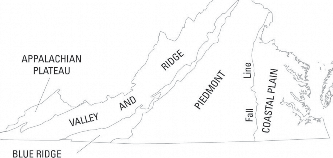
Although found throughout Virginia, corrosive water is more commonly found in the Blue Ridge and parts of the Piedmont physiographic provinces, as seen in figure 2. In some parts of far Southwest Virginia, acid mine drainage may also reduce pH, increasing the corrosiveness of groundwater.
Testing for Corrosive Water
The pH of your water is the primary factor that determines its corrosiveness. The EPA recommends that drinking water be noncorrosive, within a pH range of 6.5 to 8.5. Testing for pH can be done on-site by a trained water treatment professional or may be determined by collecting a water sample and submitting it to a certified water testing laboratory. The Virginia Household Water Quality Program maintains a list of certified laboratories on its “Resources” page at www.wellwater.bse.vt.edu/resources.php. Carefully follow all sample collection instructions. You can also contact your local Virginia Cooperative Extension (VCE) office about getting your water tested.
Other characteristics of your water — like the amount of dissolved oxygen, carbon dioxide, and hydrogen sulfide — can affect pH. In addition, the hardness, amount of total dissolved solids, alkalinity, and temperature of the water can be used to assess what is called a “saturation index.” A saturation index (SI) is a way to estimate the corrosivity of water.
Some labs report the Langelier Saturation Index (LSI). LSI values less than minus one (-1) indicate corrosive water, while LSI values more than positive one (+1) indicate noncorrosive water.
Another commonly reported index is the Ryzner Stability Index (RSI). An RSI greater than about 6.5 indicates water that is potentially corrosive, with higher values being more corrosive. If your household water is corrosive, it is strongly recommended that you test for metals like lead and copper.
Solving the Problem
Because corrosive water affects the entire household plumbing system, treatment devices like acid-neutralizing filters and chemical feed systems — which work by adding alkaline chemicals to the water to increase pH — are typically installed where the water enters the house (point-of-entry or POE). Acid-neutralizing filters consist of a corrosion-resistant tank filled with calcite (calcium carbonate in the form of limestone or marble chips) or a mixture of calcite and magnesium oxide (also called corosex). As the water flows through these materials, they are dissolved and increase the pH of the water. Depending on the mix of materials used in the acid-neutralizing filter, these devices can treat water with a pH as low as 5.3.
Chemical feed systems that use a metering pump to add an alkaline material like soda ash (sodium carbonate) to the water can also be used to increase pH for water with a pH below 5.3, but these systems are more complex and require significant regular maintenance. For this reason, chemical feed systems are used less frequently in household applications.
As with any water treatment device, it is critical that the device be maintained according to manufacturer specifications. Failing to do so can result in the device not functioning as intended. Acid-neutralizing treatment should be employed as the primary treatment option for drinking water only if the homeowner is assured that the water entering the home is otherwise pollution- and metal-free.
If, for some reason, effective pH adjustment of your water supply is not possible, consider replacing your plumbing with corrosion-resistant plumbing materials like PVC, CPVC, PEX, or other plastic plumbing materials tested by the National Sanitation Foundation (NSF) and stamped with “NSF-PW” (potable water) or “NSF-61.”
Replacing metal plumbing can minimize the effects of corrosive water. It is important to note that changing plumbing materials does nothing to change the water’s corrosiveness, but it may reduce the risk of certain metals dissolving into your drinking water. Also keep in mind that, if untreated, corrosive water will continue to corrode the metal components in water heaters and other water-using appliances.
In some cases, polyphosphates can be used to stop corrosion. Polyphosphates that are added to the water will provide a thin protective layer on the pipe surface that minimizes metal release. Polyphosphates can be applied using a cartridge or pump. Polyphosphates need a small amount of hardness to work.
If effective pH adjustment cannot be achieved and replacing metal plumbing components is not possible or practical, alternative options to reduce the concentration of lead and copper in your water include (1) flushing the plumbing system (letting the water run) before using the water for consumption, and (2) installation of a point-of- use treatment device that can remove metals.
The amount of dissolved lead and copper in the water increases the longer the corrosive water is in contact with metal plumbing and fixtures. Flushing your plumbing system of water that has been in contact with the plumbing for longer than one hour can reduce the metal concentration in the water. The time needed for flushing depends on your plumbing system. A good rule of thumb is to let the water run until it becomes as cold as it will get. You can conserve water by flushing the plumbing in the morning while showering or bathing or while doing laundry.
Be sure to also flush the kitchen faucet, the one most typically used when preparing meals or beverages, and always use cold water for drinking and cooking. After flushing the system, fill a container with water. This water can then be used for drinking and cooking throughout the day.
If water testing indicates that excessive levels of lead or copper persist after flushing, or if flushing is not an option, another alternative is a point-of-use treatment device. Suitable point-of-use treatment devices that remove lead and copper include water distillation, reverse osmosis, and activated alumina filters. Point-of-use devices typically only treat water going to a single faucet, meaning water from other faucets in the house may not be safe to drink. The devices are typically only installed on the cold water line and must be strictly maintained as instructed by the manufacturer to function properly. Note that granular activated carbon filters are not recommended for lead or copper removal.
Finally, if you have corrosive water, consider setting your water heater no higher than 140 degrees Fahrenheit. Corrosivity increases with increasing water temperature.
Summary
Consuming corrosive water is not in itself dangerous. Consuming metals, such as lead or copper that may be dissolved by corrosive water from plumbing, plumbing fixtures, appliances, or groundwater sources may pose a health risk. In high enough concentrations, dissolved metals can cause health problems, nuisance effects, or both.
If a water test indicates that you have corrosive water, there are effective measures that can be used to adjust or correct the pH of your water. The EPA recommends that drinking water be noncorrosive and be within a pH range of 6.5 to 8.5. Consult a reputable water treatment professional.
For more information on water testing and treatment and issues related to corrosive water, see the following websites:
Virginia Household Water Quality Program: www.wellwater.bse.vt.edu/resources.php.
Virginia Cooperative Extension: http://pubs.ext.vt.edu/category/home-water-quality.html.
Acknowledgments
The authors wish to thank the following individuals who reviewed this publication: Scott Reiter, agriculture and natural resources agent, VCE Prince George County Office; Dana Rizzo, Extension educator, water quality, Pennsylvania Cooperative Extension Westmoreland County Office; Sharon Skipton, Extension water quality educator, Nebraska Cooperative Extension; and Carl Zipper, associate professor, Crop and Soil Environmental Sciences, Virginia Tech.
Resources
Hairston, James E., Leigh Stripling, and John M. Beck. 2001. Corrosion. Alabama Cooperative Extension System, Alabama A&M and Auburn Universities. http://aces.edu/waterquality/articles/0311001/0311001.pdf.
Oram, Brian. 2007. Corrosion, Saturation Index, Balanced Water in Drinking Water Systems. Wilkes University, Environmental Engineering and Earth Sciences Department, Center for Environmental Quality. www.water-research.net/corrosion.htm.
Skipton, S., B. Dvorak, W. Woldt, and S. Drda. 2008. Drinking Water: Copper. University of Nebraska-Lincoln Extension Publication G1360. http://ianrpubs.unl.edu/epublic/live/g1360/build/g1360.pdf.
Swistock, B. R., W. E. Sharpe, and P. D. Robillard. 2001. Corrosive Water Problems. Penn State College of Agricultural Sciences, Cooperative Extension Publication F137. http://extension.psu.edu/water/resources/publications/water-pollutants/pollutants/corrosivewater.pdf/view.
U.S. Environmental Protection Agency (EPA). 2004. Lead and Copper Rule: A Quick Reference Guide. EPA 816-F-04-009. http://water.epa.gov/lawsregs/rules-regs/sdwa/lcr/upload/2004_03_11_lcrmr_pdfs_qrg_lcmr_2004.pdf.
U.S. Environmental Protection Agency. 2010. Secondary Drinking Water Regulations: Guidance for Nuisance Chemicals. EPA 816-F-10-079. http://water.epa.gov/drink/contaminants/secondarystandards.cfm.
Vendrell, Paul F., and Jorge H. Atiles. 2003. Your Household Water Quality: Corrosive or Scaling Water. University of Georgia Cooperative Extension Service, Circular 858-9. www.fcs.uga.edu/pubs/PDF/HACE-858-9.pdf.
Virginia Department of Environmental Quality. Physiographic Provinces of Virginia. Virginia DEQ Web Page: http://deq.virginia.gov/gwpsc/geol.html.
Virginia Cooperative Extension materials are available for public use, reprint, or citation without further permission, provided the use includes credit to the author and to Virginia Cooperative Extension, Virginia Tech, and Virginia State University.
Virginia Cooperative Extension is a partnership of Virginia Tech, Virginia State University, the U.S. Department of Agriculture (USDA), and local governments, and is an equal opportunity employer. For the full non-discrimination statement, please visit ext.vt.edu/accessibility.
Publication Date
December 16, 2024



